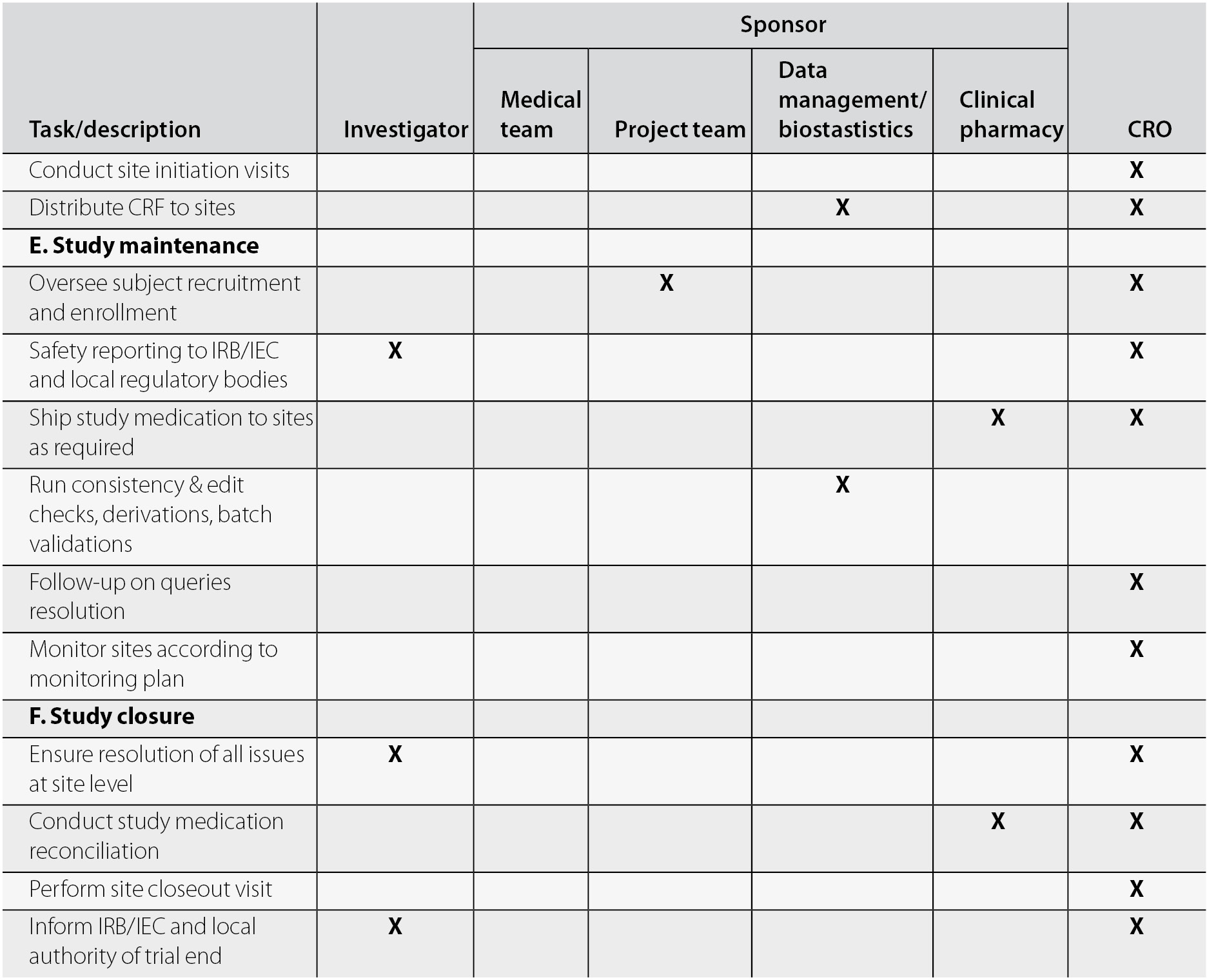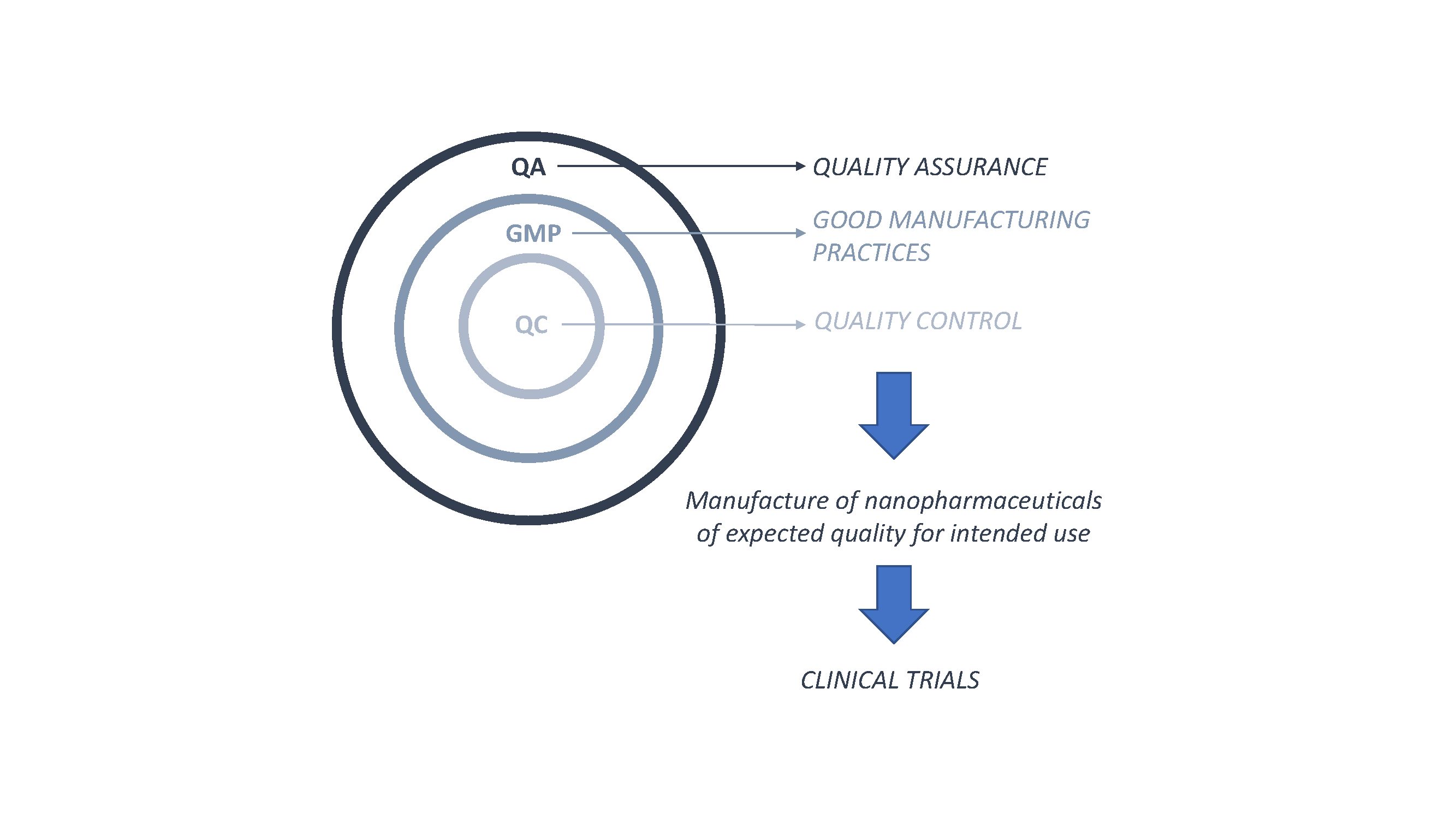
Pharmaceutics | Free Full-Text | Nanopharmaceutics: Part I—Clinical Trials Legislation and Good Manufacturing Practices (GMP) of Nanotherapeutics in the EU | HTML
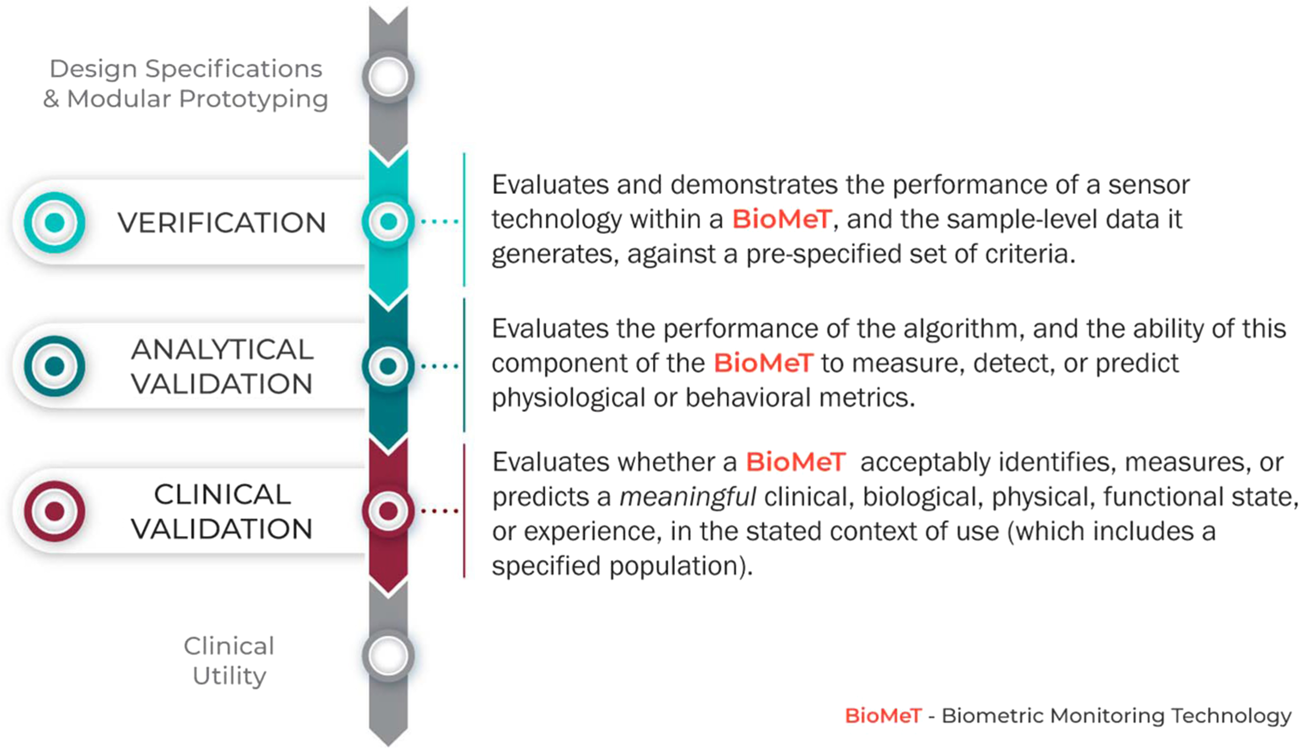
Verification, analytical validation, and clinical validation (V3): the foundation of determining fit-for-purpose for Biometric Monitoring Technologies (BioMeTs) | npj Digital Medicine

PDF) Central Institutional Ethics Committee needed to facilitate timely review of multicenter clinical trials

PDF) Central Institutional Ethics Committee needed to facilitate timely review of multicenter clinical trials

Regulatory oversight of cell therapy in China: Government's efforts in patient access and therapeutic innovation - ScienceDirect