PDF) Quality assurance within the scope of Good Clinical Practice (GCP)-what is the cost of GCP-related activities? A survey within the Swedish Association of the Pharmaceutical Industry (LIF)'s members | Karin Eriksson -

PDF) Good Clinical Practice and Audits for Dual X-ray Absorptiometry and X-ray Imaging Laboratories and Quality Assurance Centers Involved in Clinical Drug Trials, Private Practice, and Research | Barbara Blunt - Academia.edu

Principles of Good Clinical Practice (GCP) – What is it all about and who is responsible for adherence? GCP and QA All SIAC Call Mar 14, 2008 Munish Mehra, - ppt download

Clinical Trials Audit Preparation: A Guide for Good Clinical Practice (GCP) Inspections: 9780470248850: Medicine & Health Science Books @ Amazon.com

Very Helpful Resource: Good Clinical Practice: A Question & Answer Reference Guide 2018 — Clinical Pathways

PDF) Good Clinical Practice Training: Identifying Key Elements and Strategies for Increasing Training Efficiency

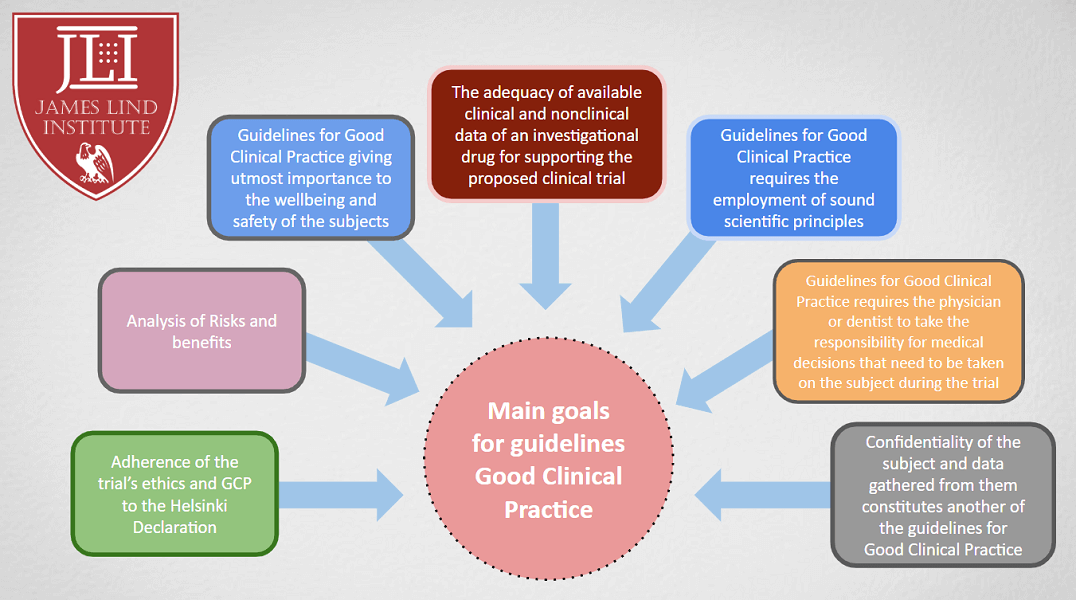
PDF) The importance of Good Clinical Practice guidelines and its role in clinical trials | Anushya Vijayananthan - Academia.edu

Good Clinical Practice A Question Answer Reference Guide Pdf - Fill Online, Printable, Fillable, Blank | pdfFiller
UNE EN ISO 14155:2021 Clinical investigation of medical devices for human subjects - Good clinical practice (ISO 14155:2020) - European Standards
Good Clinical Practice and the Evaluation of Treatment: A Response to Seto et al - W.L. Marshall, L.E. Marshall, 2008

Good Clinical Practices Concepts and Case Studies Kim Isaacs | PDF | Institutional Review Board | Clinical Trial