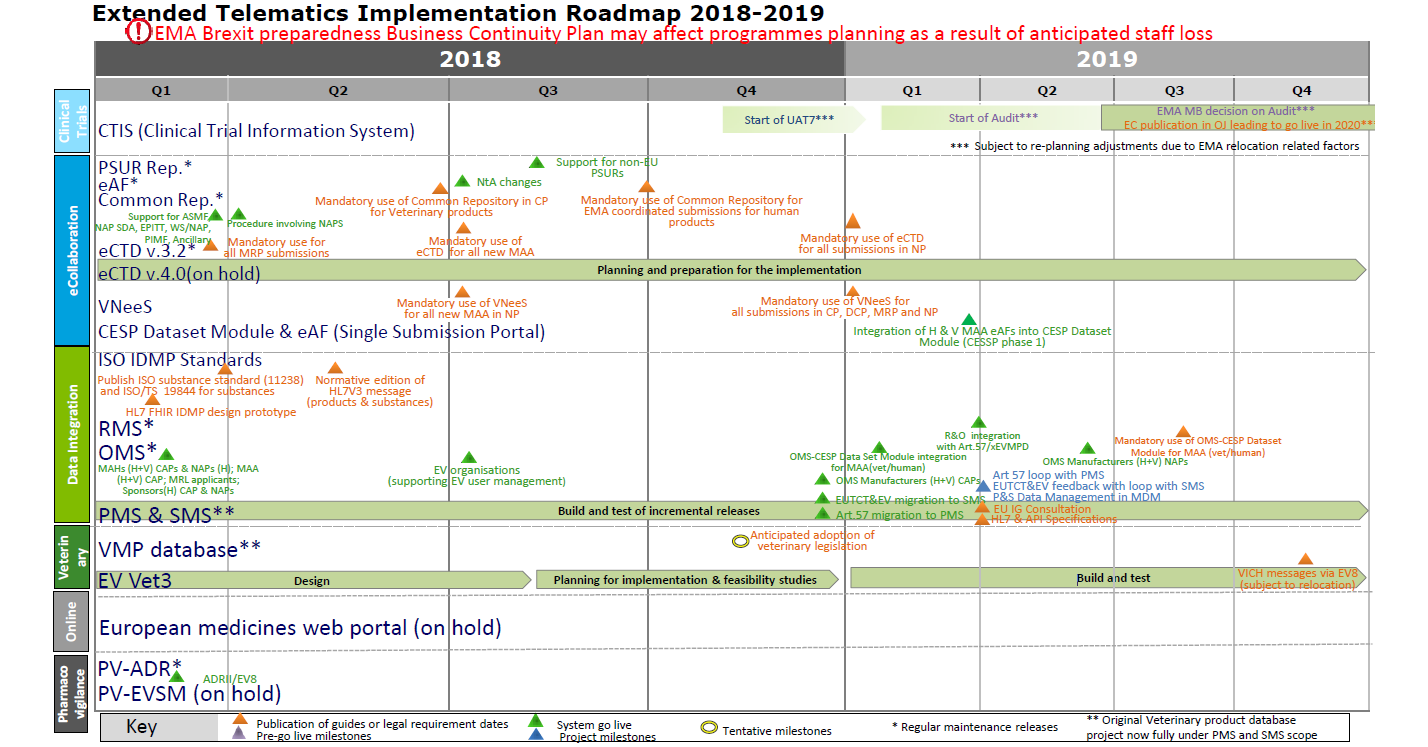
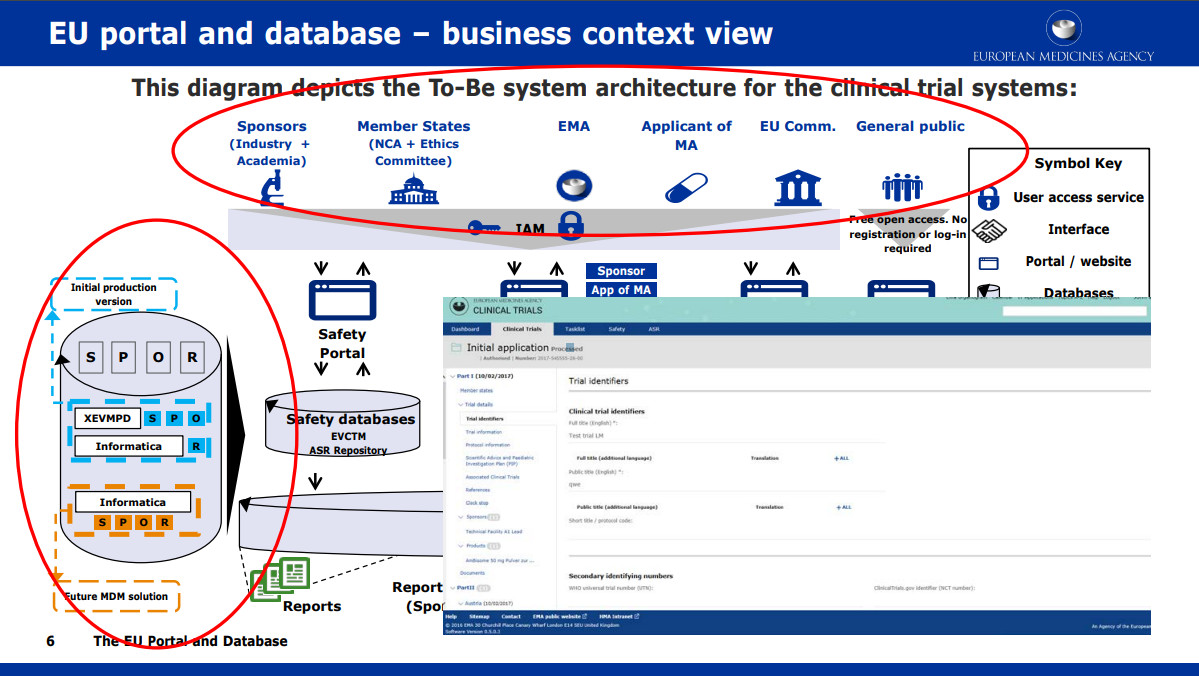
Assessment of the Regulatory Dialogue Between Pharmaceutical Companies and the European Medicines Agency on the Choice of Noninferiority Margins - Clinical Therapeutics

EU Medicines Agency on Twitter: "In a letter published today, @EU_Commission, #EMA and the Heads of Medicines Agencies remind all sponsors of #ClinicalTrials conducted in the 🇪🇺 to make results of concluded

Van Bael & Bellis auf Twitter: „EU Clinical Trial Portal and EU Database Declared Functional by EMA Board - EU Clinical Trials Regulation Expected to Take Effect on 31 January 2022

Cochrane supports European regulators as they urge clinical trial sponsors to share their results | Cochrane