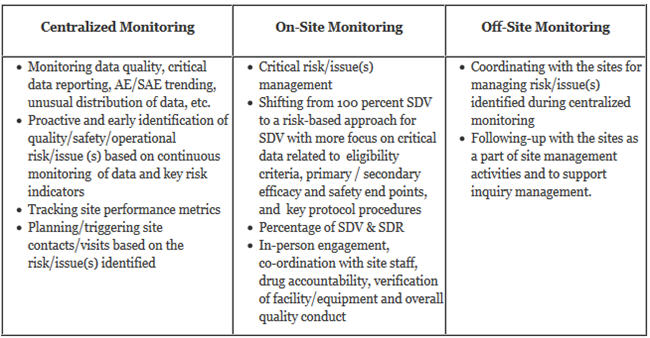
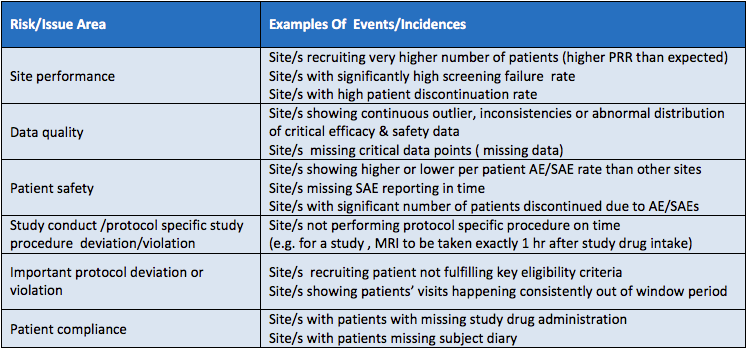
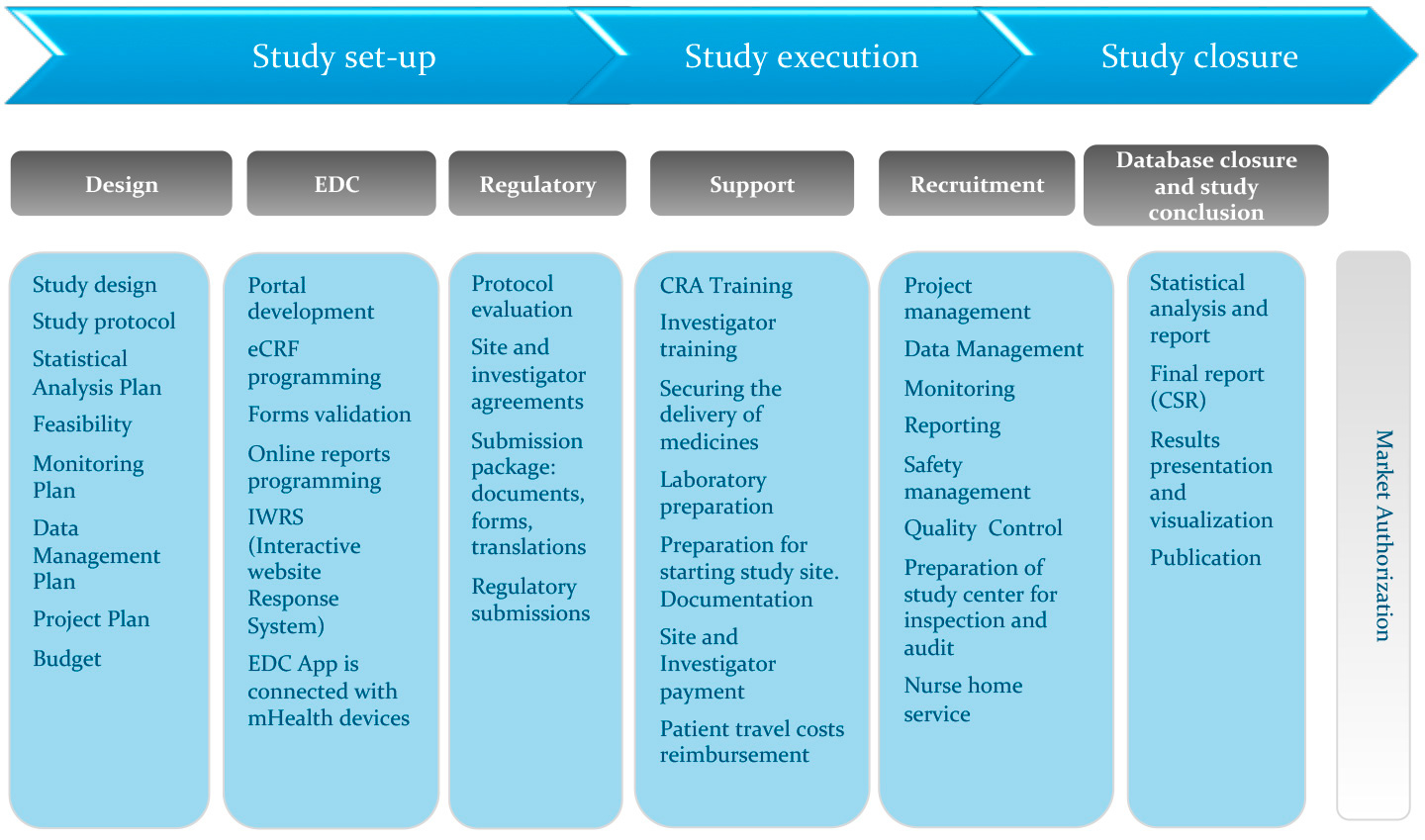
A Centralized Monitoring Approach Using Excel for the Quality Management of Clinical Trials | Applied… | How to plan, Sorority recommendation letter, Support letter
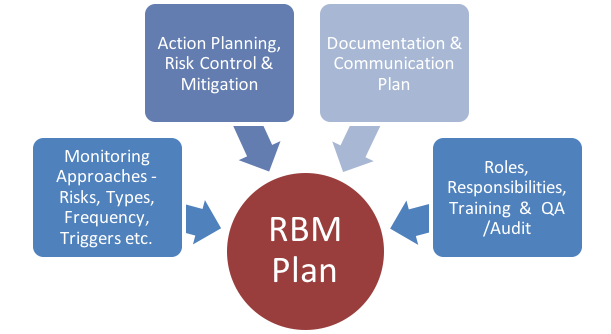
Guidelines for Developing a Data and Safety Monitoring Plan | National Institute on Drug Abuse (NIDA)

CHANGE OF MONITOR AT STUDY SITE No: CM Effective Date: 15 April 2013 Version Date: 15 March PDF Free Download

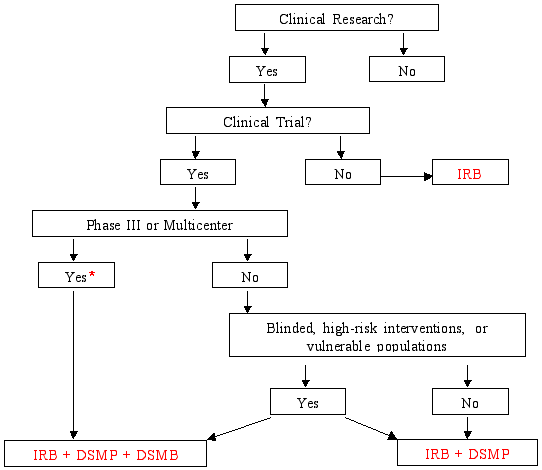
Figure 3 from Validation of a risk-assessment scale and a risk-adapted monitoring plan for academic clinical research studies--the Pre-Optimon study. | Semantic Scholar

Your Clinical Trial Monitoring Plan: 5 Tips from a Regulatory Specialist - Clinical Research IO - CRIO
Pilot, double-blind, randomized, placebo-controlled clinical trial of the supplement food Nyaditum resae® in adults with or without latent TB infection: Safety and immunogenicity | PLOS ONE

Monitoring Report Template Clinical Trials (2) - TEMPLATES EXAMPLE | TEMPLATES EXAMPLE | Report template, Treatment plan template, Clinical trials



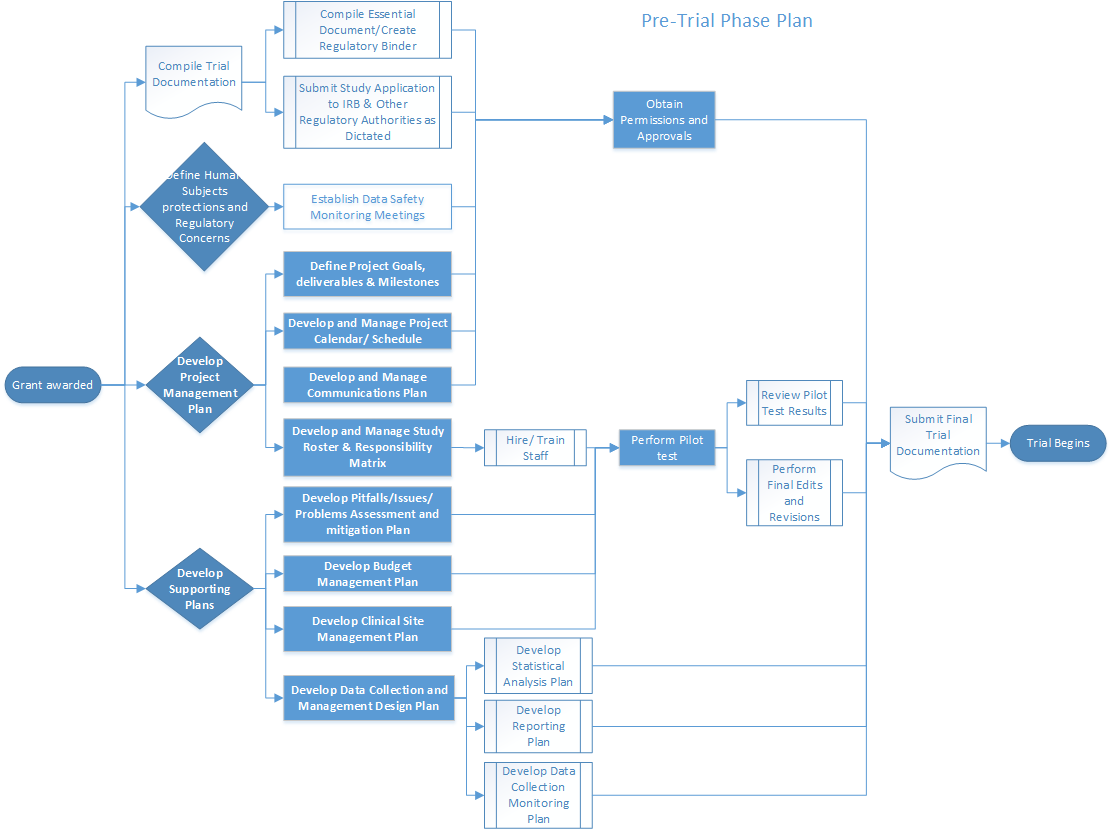
![CLINICAL DATA MONITORING PLAN (CDMoP) PROTOCOL # [0000] [TITLE] - PDF Free Download CLINICAL DATA MONITORING PLAN (CDMoP) PROTOCOL # [0000] [TITLE] - PDF Free Download](https://docplayer.net/docs-images/44/18344673/images/page_2.jpg)